Exploring New Techniques for Delivering Cancer Treatments
Cancer care is evolving beyond new drugs alone. Advances in how therapies reach tumors—from targeted carriers and implanted pumps to image-guided radiation—aim to improve precision while limiting side effects. This overview explains how delivery innovations are shaping care and research in the United States.
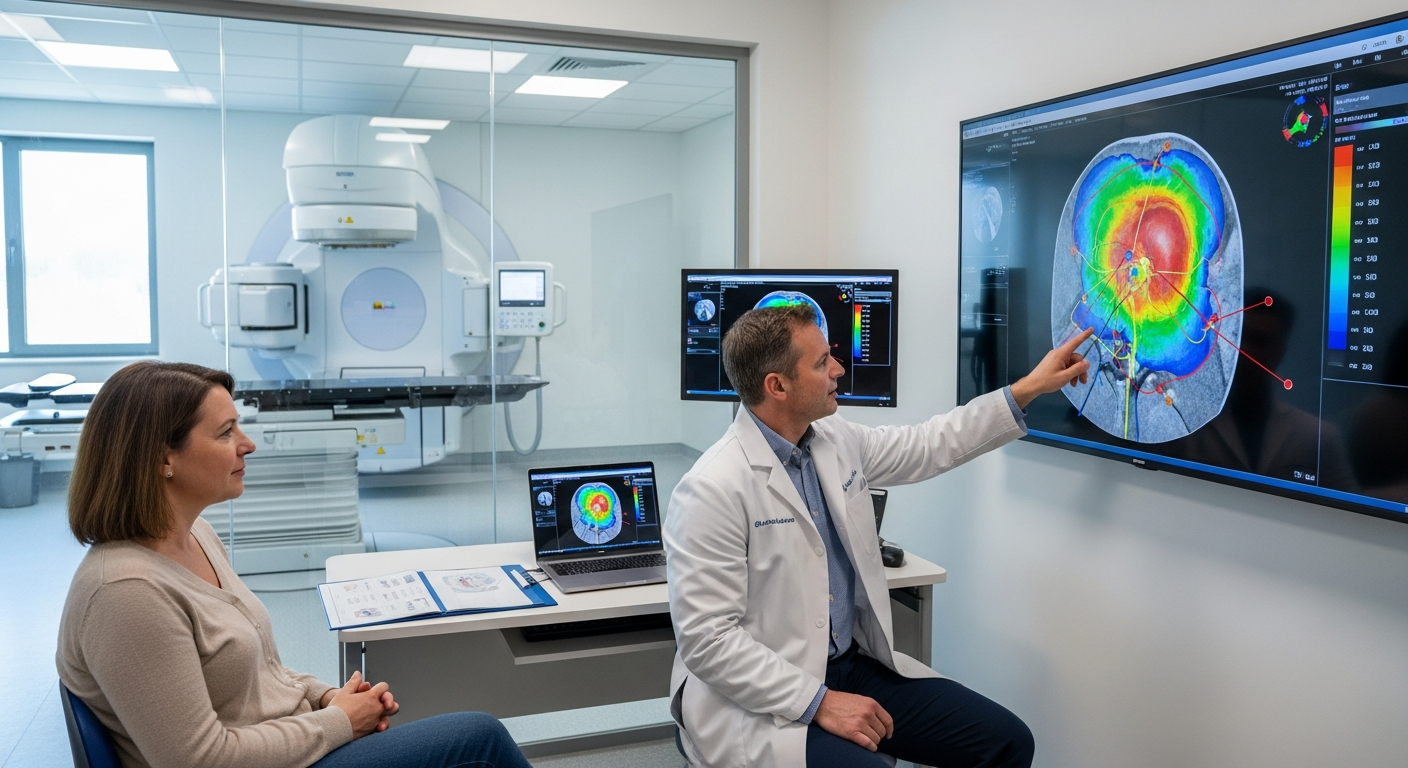
How a therapy reaches a tumor can be as crucial as the drug itself. Modern oncology increasingly focuses on delivery: getting the right dose to cancer cells while protecting healthy tissue. From smart drug carriers and localized infusions to precision radiation and intratumoral injections, delivery innovations are changing how treatments are planned and administered in the United States. These advances also reflect a broader shift toward personalization, integrating biomarkers, imaging, and device-assisted methods to match treatment routes to each patient’s needs.
This article is for informational purposes only and should not be considered medical advice. Please consult a qualified healthcare professional for personalized guidance and treatment.
What’s new in treatment delivery?
A wave of research is testing new ways to move therapies safely and precisely through the body. Nanomedicine approaches, including liposomal formulations and polymer-based nanoparticles, are designed to shield drugs in the bloodstream and release them near tumors. Antibody–drug conjugates link potent chemotherapy payloads to antibodies that recognize tumor markers, aiming to concentrate effects where they matter most. Researchers are also refining prodrugs that activate in the tumor microenvironment, such as in areas with characteristic enzymes or acidity.
For hard-to-reach sites like the brain, focused ultrasound with circulating microbubbles is being studied to temporarily open the blood–brain barrier, potentially enhancing drug entry. Convection-enhanced delivery uses small catheters to push drugs directly into brain tissue under image guidance, seeking broader local coverage than a simple injection. In the abdomen, heated chemotherapy during surgery (often called intraperitoneal hyperthermic therapy) circulates drugs directly where disease is present, while hepatic artery infusion pumps deliver high concentrations to liver tumors via the liver’s arterial supply. Across these strategies, the common goal is to increase on-target exposure and reduce systemic toxicity.
Understanding the latest delivery advances
Understanding the latest in cancer therapy delivery starts with pharmacology. How a drug is absorbed, distributed, metabolized, and excreted strongly influences which route—oral, intravenous, intratumoral, intrathecal, or regional infusion—offers the best trade-off between benefit and risk. Although the idea that tumors naturally accumulate nanoparticles (sometimes called the enhanced permeability and retention effect) has guided design, evidence shows this varies widely among tumor types and even between patients, underscoring the role of imaging and biomarkers to inform choices.
Radiation oncology provides another lens on delivery innovation. Image-guided techniques, including stereotactic body radiation therapy and pencil-beam proton therapy, aim to sculpt dose around organs at risk. Brachytherapy places sealed sources close to or inside a tumor for a steep dose fall-off, potentially limiting collateral damage. In parallel, immunotherapy delivery is diversifying: intratumoral injections of immune-stimulating agents, oncolytic viruses designed to replicate in cancer cells, and engineering of cell therapies that may be infused systemically or, in some protocols, administered regionally to concentrate activity. Each method brings practical considerations—device placement, scheduling, and monitoring—that shape real-world use in U.S. clinics and academic centers.
Methods for effective treatment delivery
Methods for effective delivery of cancer treatments combine biology with logistics. Ports and catheters support repeated intravenous therapy, while reservoirs enable intrathecal delivery to cerebrospinal fluid when the central nervous system is involved. Image guidance helps position needles and catheters with millimeter accuracy for intratumoral or regional administration. Digital tools—such as electronic reminders for oral oncolytics or connected pill caps—aim to support adherence, which is essential for therapies that rely on consistent exposure.
Selecting the delivery route also depends on tumor location, size, and vascularity, prior therapies, and patient-specific factors like organ function and mobility. In the United States, multidisciplinary tumor boards often weigh these variables alongside clinical trial eligibility. For some cancers, combining systemic therapy with localized delivery can improve symptom control or shrink tumors to enable surgery or radiation. In all cases, safety monitoring—blood counts, organ tests, and imaging—guides dose adjustments and timing to keep treatment on track while limiting adverse effects.
Practical considerations and safety
Innovative delivery approaches can introduce new risks and workflow needs. Implantable pumps and reservoirs require surgical placement and maintenance, and they can fail or become infected. Regional chemotherapy may cause localized side effects distinct from systemic regimens. Intratumoral strategies require careful imaging and planning to ensure that enough of the tumor volume is covered. When the brain or spine is involved, teams evaluate neurological risks and coordinate peri-procedural care. For radiation, planning scans, motion management, and daily image verification are central to delivering precise dose safely.
Equity and access also influence delivery choices. Some techniques are available primarily at large academic centers or through clinical trials, which may require travel. Insurance coverage can vary by plan, particularly for device-based and procedure-intensive approaches. Supportive care services—in patient homes or through local services in their area—help many people manage side effects and logistics between visits, an often-overlooked part of effective delivery.
What to watch as research evolves
As research progresses, several themes stand out. First, better patient selection—using molecular profiling, advanced imaging, and circulating biomarkers—may improve who benefits from specific delivery routes. Second, combination strategies are likely to expand, pairing localized treatments with systemic immunotherapy or targeted agents. Third, real-world data from U.S. health systems will clarify which approaches deliver durable outcomes outside of tightly controlled trials.
Standardization is also developing: consistent manufacturing for complex therapeutics, clearer device protocols, and interoperable imaging and planning tools. Safety frameworks are maturing, including monitoring for rare but serious immune or neurological events tied to certain delivery methods. Ultimately, the success of any approach rests on balancing precision with practicality, aligning science with what patients can reasonably access and sustain over time.
In summary, delivery matters as much as the medicine itself. From nanoparticle carriers and antibody–drug conjugates to intratumoral injections, focused radiation, and regional infusions, the field is expanding options to concentrate therapy where it is needed and limit exposure elsewhere. The most suitable route depends on diagnosis, tumor biology, and individual circumstances, with ongoing studies in the United States continuing to refine what works best and for whom.




